CAMBRIDGE, MA, 8-Oct-2025 — /EuropaWire/ — CREATE Medicines, Inc. (formerly Myeloid Therapeutics, Inc.) today announced an expanded strategy to move beyond myeloid-focused in vivo programming toward multilineage immune programming, including T cells, myeloid cells, and NK cells, to deliver scalable, redosable, off-the-shelf therapies with greater clinical impact.
CREATE is the most advanced clinical-stage company in RNA-based in vivo CAR therapeutics, with programs targeting HER2, TROP2, and GPC3-positive solid tumors and validated targets for in vivo CAR-T mediated B cell depletion. Building on this foundation, CREATE Medicines is pioneering a new era in multi-immune programming with an expanded platform designed to deliver transformative therapies across oncology, autoimmunity, and fibrosis.
“Our clinical work in more than 40 patients has proven that we can tolerably and repeatedly program immune cells inside the body,” said Daniel Getts, Ph.D., Chief Executive Officer of CREATE Medicines. “We are now extending those capabilities to program multiple immune lineages for deeper, more durable responses, starting with MT-304, our HER2 multi-immune CAR in breast cancer. In parallel, we are advancing the first-ever RNA retrotransposon-based in vivo CAR-T for B-cell depletion in oncology and autoimmunity.”
Clinical Validation
CREATE Medicines has generated one of the largest clinical datasets in the in vivo CAR field, with more than 40 patients treated across multiple Phase 1 studies. These trials provide not only proof-of-mechanism but also critical insights that de-risk next generation multi-cell programs.
- Proof-of-mechanism: Paired biopsies confirmed CAR+ immune cells infiltrating tumors, with immune remodeling and CD8 T cell recruitment.
- Safety and repeat dosing: Over 200 doses delivered with a consistent, manageable safety profile and no cumulative toxicities.
- Evidence of activity: CAR expression detected in circulating immune cells, with stable disease in several patients and a confirmed partial response on treatment for 16 months.
These results validate CREATE’s ability to reprogram immune cells inside the body and provide the foundation for expansion into multi-lineage programming across T cells, myeloid cells, and NK cells.
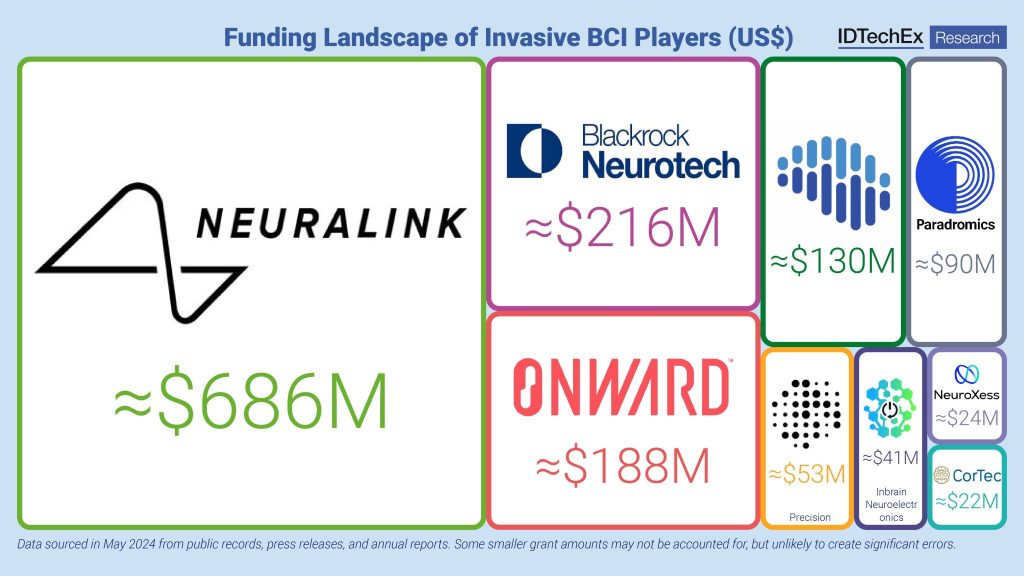
CREATE is strongly supported by a syndicate of leading life science investors, including Newpath Partners, ARCH Venture Partners, 8VC, and Hatteras Venture Partners. These firms share the company’s vision to transform immunotherapy through in vivo multi-immune programming and remain highly committed to its continued success.
Pipeline Updates and Anticipated Milestones
- MT-302 (TROP2; solid tumors): Dose escalation completed; tolerable safety profile.
- MT-303 (GPC3; hepatocellular carcinoma): Dose escalation ongoing.
- MT-304 (HER2; solid tumors): First patient expected Q4 2025; first-in-class multi-immune CAR engaging NK and myeloid cells.
- Retrotransposon-based in vivo CAR-T: First all-RNA product candidate with permanent CAR integration for B-cell depletion.
- Additional multi-lineage programs in preclinical development across oncology and immunology.
Platform Differentiation
At the core of CREATE’s platform is targeted, programmable cell engagement:
- Selective activation: Cell-specific receptors and LNPs for precise T, myeloid, and NK cell programming.
- Flexible durability: Transient or stable CAR expression, with permanent integration via RNA-based retrotransposon technology.
- Industry-leading RNA: Up to 8+ days of linear mRNA expression with no reactogenicity.
- Speed and scalability: Concept-to-clinic in <12 months with low-cost manufacturing.
SOURCE: EuropaWire